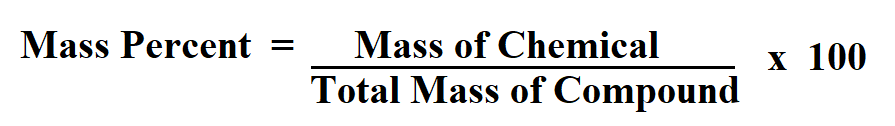
Step 2: Determine the compound’s total mass. To express the value as a percentage, you need to multiply by 100 at the top. We perform the calculation and determine that the percent by mass of the active ingredient in this ointment is 2.2 percent. Mass percent (mass of chemical / total mass of compound) x 100 is the formula for the mass percent of a compound. Calculate the percent by mass of each element in sodium nitrate (NaNO3). Mass Percent (Mass of Solute / Mass of Solution)100 Mass (m solute / mSolution)100 This formula uses 3 Variables Variables Used Mass Percent - Mass Percent is a way of expressing a concentration or describing the component in a particular mixture. Make sure that the mass units match before solving. Hint: First find the mass of each, then find each elements percent of the total. In simple terms, the formula for mass percentage of an element or the percent composition by mass is given as: Percent Composition by Mass (Mass of element / Molecular weight of compound) 100 Typically, the mass or weight of the elements and the compound is taken in grams. We can substitute the mass of the active ingredient and the mass of the anti-inflammatory ointment into the key equation. Mass percent (mass of chemical / total mass of compound) x 100 is the formula for the mass percent of a compound. In this problem, the main active ingredient is the solute, and the anti-inflammatory ointment is the solution. The solvent is the substance doing the dissolving, and the solution is a mixture produced by combining the solute and solvent. The solute is the substance that is dissolved. 
Enter a valid molecular formula and press the calculate button to determine. To use this equation, we should recognize a few key terms. Determine the Molar mass and Percent mass composition of a linear chemical formula. In order to find the percent by mass of the active ingredient in this ointment, we must recognize the key equation: percent concentration by mass is equal to the mass of the solute divided by the mass of the solution times 100 percent. What is the percent by mass of the active ingredient in this ointment? The same measurement techniques as step 1 should be used.įor this example, the mass of the entire solution/substance is found to be 150kg.įinally, calculate the mass percent using the values of the compound and substance found above.50 grams of anti-inflammatory ointment was found to contain 1.1 grams of the main active ingredient. (a) Determine the empirical formula of the compound (b) Determine the molecular formula for this compound, given that its molecular weight is 152.0 g mol 1. This is the mass of the entire solution or substance including the compound being analyzed. The percentage mass of nitrogen in one of the oxides is 36.85. Next, measure the mass of the entire substance. Use accurate calculations of scales for this.įor this example, the mass of the compound is measured to be 50 kg. This should be only the mass of the compound of material you are trying to calculate the mass percent for. Mass Percent Exampleįirst, measure the mass of the compound. Explanation: To calculate the mass percent, calculate the the individual masses. For a solution, the mass percent is expressed. Mass Percent DefinitionĪ mass percent is a ratio of the total mass of a singular compound within a larger mass of a substance. Calculate the mass percent of sulfur in sulfuric acid displaystyle (H2SO4). Mass percent is used as a way of expressing a concentration or a way of describing a component in a mixture. To calculate a mass percent, divide the total mass of the compound by the mass of the substance, then multiply by 100. percent v/v solution is calculated by the following formula using the milliliter as the base measure of volume (v): v/v mL of solute/100 mL of solution.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
